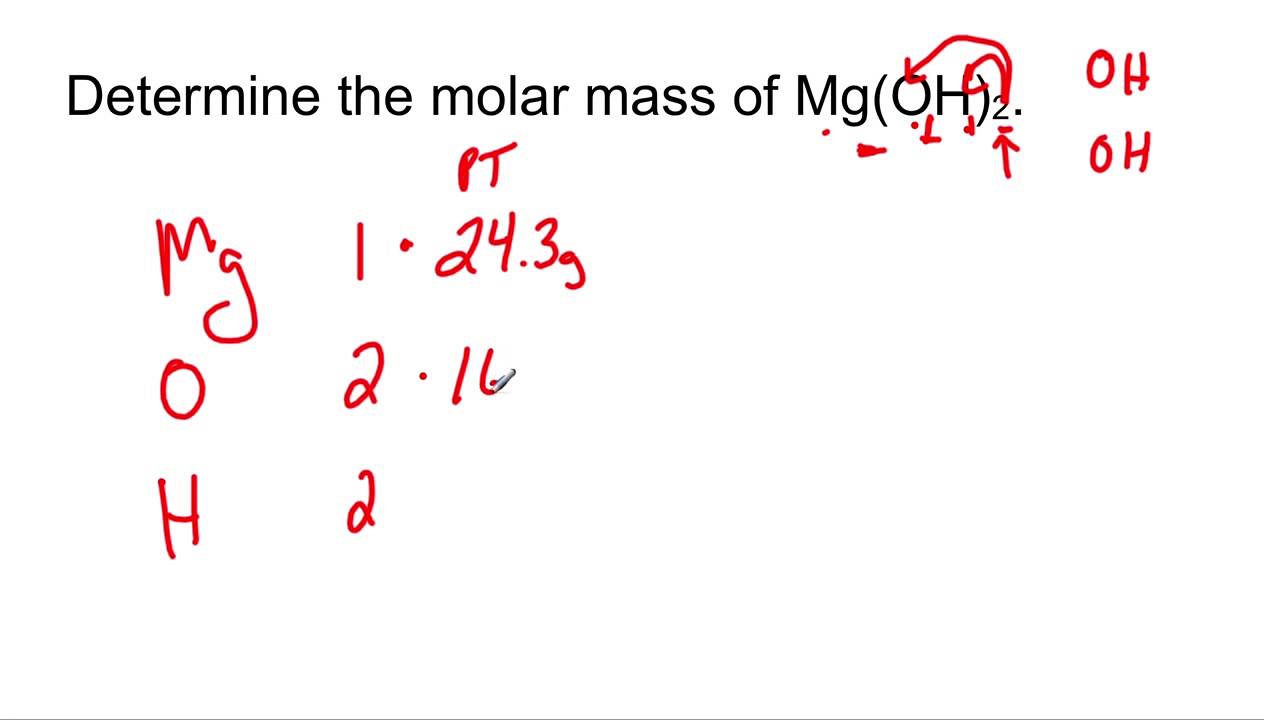
Consider, for example, mixing aqueous solutions of the soluble compounds sodium carbonate and calcium nitrate. It is important to realize that this equilibrium is established in any aqueous solution containing Ca 2+ and CO 3 2– ions, not just in a solution formed by saturating water with calcium carbonate. Solution ( a ) AgI ( s ) ⇌ Ag + ( a q ) + I − ( a q ) K sp = ( b ) CaCO 3 ( s ) ⇌ Ca 2+ ( a q ) + CO 3 2− ( a q ) K sp = ( c ) Mg ( OH ) 2 ( s ) ⇌ Mg 2+ ( a q ) + 2OH − ( a q ) K sp = 2 ( d ) Mg ( NH 4 ) PO 4 ( s ) ⇌ Mg 2+ ( a q ) + NH 4 + ( a q ) + PO 4 3− ( a q ) K sp = ( e ) Ca 5 ( PO 4 ) 3 OH ( s ) ⇌ 5Ca 2+ ( a q ) + 3PO 4 3− ( a q ) + OH − ( a q ) K sp = 5 3 ( a ) AgI ( s ) ⇌ Ag + ( a q ) + I − ( a q ) K sp = ( b ) CaCO 3 ( s ) ⇌ Ca 2+ ( a q ) + CO 3 2− ( a q ) K sp = ( c ) Mg ( OH ) 2 ( s ) ⇌ Mg 2+ ( a q ) + 2OH − ( a q ) K sp = 2 ( d ) Mg ( NH 4 ) PO 4 ( s ) ⇌ Mg 2+ ( a q ) + NH 4 + ( a q ) + PO 4 3− ( a q ) K sp = ( e ) Ca 5 ( PO 4 ) 3 OH ( s ) ⇌ 5Ca 2+ ( a q ) + 3PO 4 3− ( a q ) + OH − ( a q ) K sp = 5 3 Ĭheck Your LearningWrite the dissolution equation and the solubility product for each of the following slightly soluble compounds:ĬaCO 3 ( s ) ⇌ Ca 2+ ( a q ) + CO 3 2− ( a q ) K s p = 8.7 × 10 − 9 CaCO 3 ( s ) ⇌ Ca 2+ ( a q ) + CO 3 2− ( a q ) K s p = 8.7 × 10 − 9 (e) Ca 5(PO 4) 3OH, the mineral apatite, a source of phosphate for fertilizers (d) Mg(NH 4)PO 4, magnesium ammonium phosphate, an essentially insoluble substance used in tests for magnesium (c) Mg(OH) 2, magnesium hydroxide, the active ingredient in Milk of Magnesia (b) CaCO 3, calcium carbonate, the active ingredient in many over-the-counter chewable antacids (a) AgI, silver iodide, a solid with antiseptic properties Write the dissolution equation and the solubility product expression for each of the following slightly soluble ionic compounds: Writing Equations and Solubility Products For example, a saturated solution of silver chloride is one in which the equilibrium shown below has been established. 
A solute with finite solubility can yield a saturated solution when it is added to a solvent in an amount exceeding its solubility, resulting in a heterogeneous mixture of the saturated solution and the excess, undissolved solute. Recall from the chapter on solutions that the solubility of a substance can vary from essentially zero ( insoluble or sparingly soluble) to infinity ( miscible). This section applies previously introduced equilibrium concepts and tools to systems involving dissolution and precipitation. An understanding of the factors affecting compound solubility is, therefore, essential to the effective management of these processes. These equilibria underlie many natural and technological processes, ranging from tooth decay to water purification. Solubility equilibria are established when the dissolution and precipitation of a solute species occur at equal rates. Carry out equilibrium computations involving solubility, equilibrium expressions, and solute concentrations. 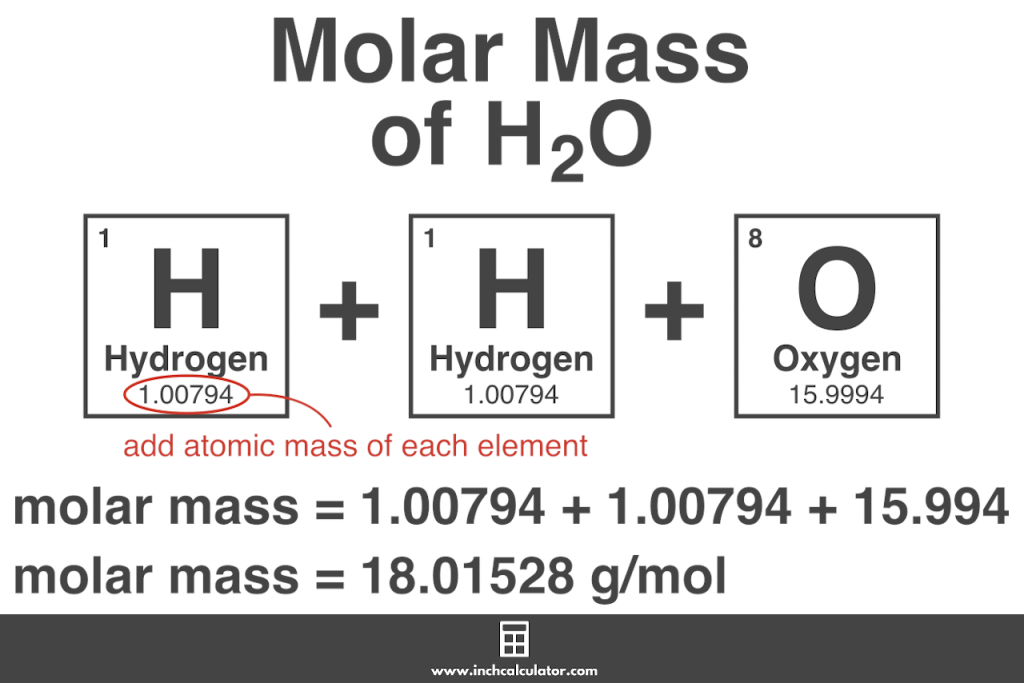
Write chemical equations and equilibrium expressions representing solubility equilibria.Its name is derived from the Greek word thallos, which means twig or green shoot.By the end of this section, you will be able to: Thallium has also been used with sulfur, selenium or arsenic to produce low melting glasses which become fluid between 125 and 150 ☌, while thallium oxide has been used to produce glasses with a high index of refraction, and is also used in the manufacture of photo cells. Thallium bromide-iodide crystals have been used as infrared optical materials. This ability makes the compound useful in photocells. Thallium is primarily used for its electrical conductivity as thallium sulfide, which changes with exposure to infrared light. Thallium is a post-transition metal that is not found free in nature. Thallium was discovered by Sir William Crookes in 1861 and first isolated by Claude-Auguste Lamy in 1862. The thallium atom has a radius of 170 pm and a Van der Waals radius of 196 pm. The number of electrons in each of thallium's shells is 2, 8, 18, 32, 18, 3 and its electron configuration is 4f 14 5d 10 6s 2 6p 1. Thallium (atomic symbol: Tl, atomic number: 81) is a Block P, Group 13, Period 6 element with an atomic weight of 204.38. Thin Film Deposition & Evaporation Materials. 
Additive Manufacturing & 3D Printing Materials.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
